High target disease prevalence, product launches, increasing strategic developments such as partnerships and agreements, favorable research funding scenario are key factors contributing to high CAGR of Cardiovascular Device Market during forecast period.
New York City, United States – June 25, 2020 /MarketersMedia/ —
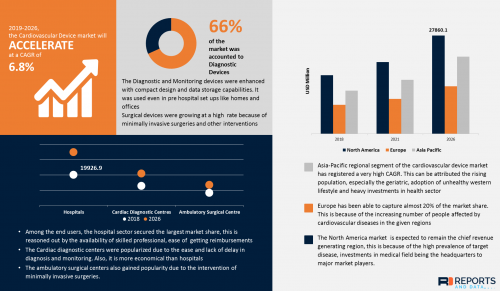
The recent study on Cardiovascular Device market by Reports and data suggests that the market was estimated to attain a value of USD 69.08 billion by the end of 2026 with a CAGR of 6.8%. The most common Cardiovascular diseases include stroke, hypertensive heart disease, rheumatic heart failure, heart arrhythmia, congenital heart disease etc. Cardiovascular Devices is an apparatus or a material made with a purpose of prevention, treatment and alleviation of the Cardiovascular disorder. It can be a device used for monitoring and diagnosis or for surgical intervention in the treatment of the diseases.
This is the latest report covering the current COVID-19 scenario. The coronavirus pandemic has greatly affected every aspect of the worldwide industry. It has brought along various changes in market conditions. The rapidly changing market scenario and initial and future assessment of the impact are covered in the research report. The report discusses all the major aspects of the market with expert opinions on the current status along with historical data.
Get a sample PDF copy of the report @ https://www.reportsanddata.com/sample-enquiry-form/1430
Key players mentioned in the research report are:
Terumo Corporation, Stryker Corporation, Abbott Laboratories, General Electric Company, Boston Scientific Corporation, Edwards Lifesciences Corporation, and W.L. Gore & Associates, Getinge Group and Becton Dickinson and Company
In terms of applications, the global Cardiovascular Devices market can be segmented into:
Coronary Heart Disease
Sudden Cardiac Arrest
Stroke
Cerebrovascular Heart Disease
Others
In terms of types, the global Cardiovascular Devices market can be segmented into:
Diagnostic and monitoring devices
Surgical Devices
The Cardiovascular Devices Market is analyzed on the basis of dynamics of demand and supply, pricing, total volume produced, revenue generated, and others. The manufacturing is studied on the basis of several factors such as manufacturing plant distribution, industry production, production capacity, and research and development. It also delivers accurate market evaluations with the use of SWOT analysis, investment analysis, returns analysis, and growth trend analysis, and others.
To get a discount on the report, click @ https://www.reportsanddata.com/discount-enquiry-form/1430
For geographical segmentation, regional supply, application-wise, and type-wise demand, key players, and others, this report covers the following regions: North America, Europe, Asia-Pacific, South America, and Middle East & Africa. The report sheds light on the competitive landscape of the market that covers the product offerings, services, market shares, and business overview. This Cardiovascular Devices Market research report covers various dynamic aspects like the market drivers, restraints and challenges, and growth prospects. The prominent and leading companies are profiled in the report.
Further key findings from the report suggest
1. Asia-Pacific regional segment of the cardiovascular device market has registered a very high CAGR. This can be attributed the rising population, especially the geriatric, adoption of unhealthy western lifestyle and heavy investments in health sector
2. Among the end users, the Cardiac diagnostic centers were popularized due to the ease and lack of delay in diagnosis and monitoring. Also, it is more economical than hospitals and hence it has registered a CAGR of 6.5%
3. Among the devices for Diagnostic and Monitoring, the ECG holds at least 40% of the share, this can be attributed to the augmented capacities of the new generation electrocardiograms.
4. The current and future product approvals have determined the strength of the market in the foreseeable future. For instance, the Medtronic plc received approval from United States Food and Drug Administration Systems (USAD) for patients with chronic and severe heart failures to be treated with a new approach like the less invasive-implant approach via thoracotomy
5. The market being consolidated and restricted to a few very strong key players, the market situation is very difficult for the entrants especially for entrants to get tax cuts and product approvals
Get the full report description, TOC, Table of Figures, Charts @ https://www.reportsanddata.com/report-detail/cardiovascular-devices-market
Thank you for reading our report. For further details or to inquire about customization, please let us know and we will offer you the report as per your needs.
About Us:
Our in-house experts assist our clients with advice based on their proficiency in the market that helps them in creating a compendious database for the clients. Our team offers expert insights to clients to guide them through their business ventures. We put in rigorous efforts to keep our clientele satisfied and focus on fulfilling their demands to make sure that the end-product is what they desire. We excel in diverse fields of the market and with our services extending to competitive analysis, research and development analysis, and demand estimation among others, we can help you invest your funds in the most beneficial areas for research and development. You can rely on us to provide every significant detail you might need in your efforts to make your business flourish.
Contact Info:
Name: John Watson
Email: Send Email
Organization: Reports And Data
Address: 40 Wall St. 28th floor New York City, NY 10005 United States
Phone: +1-212-710-1370
Website: https://www.reportsanddata.com/
Source URL: https://marketersmedia.com/cardiovascular-devices-market-to-reach-usd-6908-billion-by-2026/88965669
Source: MarketersMedia
Release ID: 88965669






